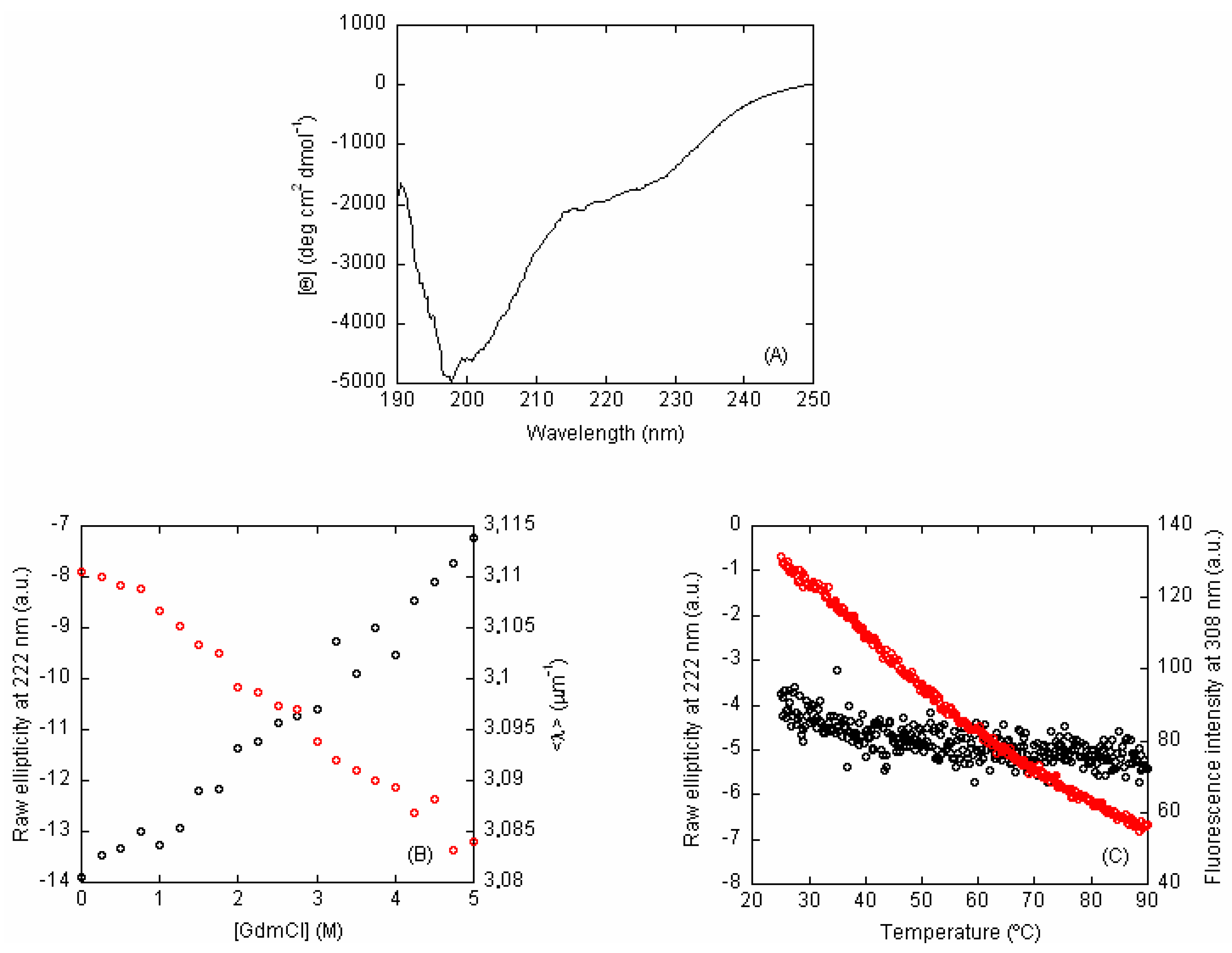
Buffer cut off wavelength for circular dichroism
At pH 3.5, HSA retained almost all of its capacity for binding to dansylglycine.

We found that HSA in its basic form at pH 9.0, which causes the protein to be less rigid, lost the capacity to bind dansylglycine. The three-dimensional structure of HSA is pH-dependent, and different conformations have been characterised. These findings suggest the relevance of the aromatic amino acids in the site II, since HOBr is a more efficient oxidant of these residues in proteins than HOCl. A comparison was made between hypochlorous acid (HOCl) and hypobromous acid (HOBr), and revealed that site II in the protein is more susceptible to alteration provoked by the latter oxidant. The induced chirality of dansylglycine was susceptive to the alteration caused by the oxidation of the protein. The intensity of the ICD signal was dependent on the temperature and revealed that the complexation between the protein and the ligand was reversible.
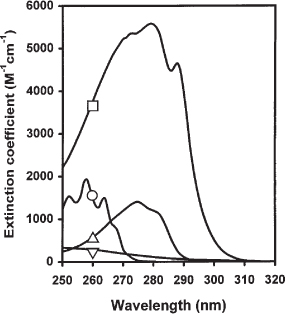
Using this ICD signal and site-specific ligands of HSA, we confirmed that dansylglycine is a site II ligand. We found that the complexation of HSA with dansylglycine resulted in the appearance of an ICD band centred at 346 nm. Here, we investigated the ICD in dansylglycine provoked by its binding to human serum albumin (HSA). Dansylglycine belongs to a class of dansylated amino acids, which are largely used as fluorescent probes for the characterization of the binding sites in albumin. Induced circular dichroism (ICD), or induced chirality, is a phenomenon caused by the fixation of an achiral substance inside a chiral microenvironment, such as the hydrophobic cavities in proteins.
